The landscape of clinical trials is undergoing a fundamental transformation. As we move through 2026, the traditional benchmarks of success for a clinical research site-once defined solely by recruitment numbers and paper-heavy documentation are being replaced by a new paradigm of Artificial Intelligence (AI) integration and high-level clinical synthesis.
The conversation around AI in clinical research has shifted. We are no longer asking if AI will be used, but how it is currently redefining the bottom line for pharmaceutical sponsors and the operational flow for clinical sites.
In 2026, the successful integration of AI is the difference between a trial that stays on budget and one that fails to launch.
1. For Sponsors: Boosting ROI through Data Intelligence
Pharmaceutical companies are facing a “yield” problem. Traditional trial models are expensive and often provide a slow Return on Investment (ROI). AI is changing this by optimizing the decision-making process before a single patient is enrolled.
• Predicting Trial Success: Using machine learning to analyze historical trial data, sponsors can now predict which protocols are likely to succeed and which recruitment targets are unrealistic.
• Optimizing Decentralized Clinical Trials (DCT’s): AI helps sponsors manage the complex logistics of DCT’s analyzing data from wearables and remote monitoring in real-time to ensure data integrity without the need for constant site visits.
• Resource Allocation: AI models can determine the exact “man-power” or investment required for a study, preventing the over-hiring or under-funding that often plagues Phase II and III trials.

2. For Clinical Sites: Revolutionizing Pre-Screening and Feasibility
At the site level, the biggest “time-thief” is feasibility. Traditionally, determining if a site can actually deliver on a protocol involves manual chart reviews and guesswork. AI is making this process scientific.
• Automated Pre-Screening: AI-powered algorithms can scan Electronic Health Records (EHRs) in seconds to identify eligible participants based on complex inclusion/exclusion criteria. This turns a weeks-long process into a daily, automated report.
• Feasibility Accuracy: By using AI to analyze local patient demographics and past performance metrics, sites can give sponsors a realistic “feasibility score.” This builds trust and ensures that the site is actually a good fit for the study.
• Enhanced Triage: AI tools allow clinical leads to triage potential participants more effectively, ensuring that the clinical team’s time is spent on high-probability candidates.

3. The Future: A Collaborative Tech-Stack
The true power of AI in clinical research lies in the synergy between the sponsor’s vision and the site’s execution. When sponsors use AI to identify the right ROI and sites use AI to identify the right patients, the entire lifecycle of a drug or device is accelerated.
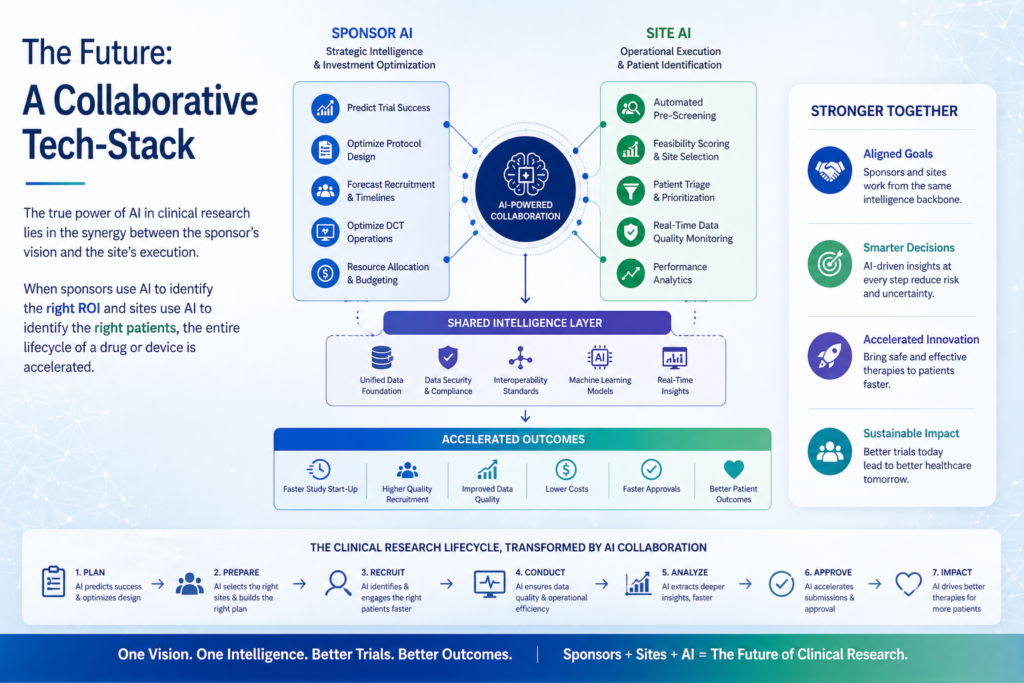
As a clinical research lead, I believe that embracing these tools isn’t about replacing human judgment it’s about empowering it. By removing the manual “noise,” we allow researchers to return to what they do best: ensuring patient safety and driving scientific innovation.
Reference:
https://www.deloitte.com/us/en/insights/topics/technology-management/tech-trends.html

Leave a Reply