The traditional clinical trial model characterized by discrete phases, manual data entry, and the long “hiatus” between database lock and regulatory review is officially entering the archives. On April 28, 2026, the FDA unveiled a landmark initiative to implement Real-Time Clinical Trials (RTCT), moving the industry toward a model of Continuous Clinical Development.
As a Clinical Research Lead currently navigating this transition, I see this shift as more than just a technological upgrade; it is a fundamental re-engineering of the investigator-regulator relationship. However, the path to accelerated drug approval is paved with significant operational and technical hurdles that require immediate strategic planning.
The April 2026 Mandate: What Site Leads Need to Know
The FDA’s recent Request for Information (RFI), open for comment until May 29, 2026, outlines a pilot program designed to assess how AI and cloud-integrated infrastructure can optimize early-phase trials. The agency has already validated the technical feasibility of this approach through proof-of-concept trials with AstraZeneca (TRAVERSE study) and Amgen (STREAM-SCLC study).
In these pilots, the FDA utilized technical intermediaries to ingest data signals in near real-time. For site leads and biotech executives, this means the “quiet periods” of a trial are disappearing.
1. The “Signal-vs-Noise” Technical Risk
The most significant technical challenge of RTCT is the compression of the quality control sequence. In a traditional workflow, data is cleaned, queried, and verified before it ever reaches a regulator’s dashboard. In a real-time stream, that buffer vanishes.
1. The Problem of Raw Data: A miscoded lab value or a duplicate adverse event entry can now look like a critical safety signal to the FDA almost immediately.
2. Regulatory Alert Fatigue: Without a defined “materiality framework,” routine site-level errors could trigger unnecessary clinical holds or inquiries, creating a feedback loop that slows development instead of accelerating it.
3. Market Volatility: For single-asset biotech firms, an unverified “preliminary signal” visible to regulators could lead to premature public disclosures or investor panic before the site even has a chance to correct the data.
2. Addressing the “Operational Drag” on Research Sites
While the technology makes the data continuous, the human workflows at the site level remain largely tethered to manual processes. As we move to RTCT, “operational drag” the cumulative friction of fragmented workflows—becomes a primary risk to trial success.
Infrastructure Requirements
Sites must now move beyond simple EMR integration. To participate in the FDA’s upcoming summer pilot, sites will likely need:
1. Automated eSource-to-EDC Pipelines: Eliminating manual transcription is no longer a luxury; it is a requirement for real-time compliance.
2. Continuous Monitoring Staffing: The role of the Clinical Research Coordinator (CRC) is shifting from periodic data entry to real-time data management and query resolution.
The “Two-Tier” System Risk
There is a growing concern among stakeholders that RTCT could create a divide in the research ecosystem. High-resource academic centers with advanced tech-stacks (like those partnered with Paradigm Health) will be the natural choice for these pilots, potentially excluding community-based sites and diverse patient populations that lack the necessary digital infrastructure.
3. How to Prepare: Strategic Recommendations
To stay competitive and compliant in this new regulatory environment, site leads and sponsors should focus on three areas:
1. Implement “Quality by Design” (QbD): Build automated validation checks directly into your eSource. If the system prevents the “noise” (errors) at the point of entry, the “signal” sent to the FDA remains clean.
2. Define Your Materiality Thresholds: Before starting an RTCT, work with the sponsor and the FDA to define what constitutes a “reportable real-time event” versus a “routine data correction.”
3. Audit Your Tech Stack: Ensure your vendors can handle Digital Health Technologies (DHTs) and continuous data streaming without creating data silos.
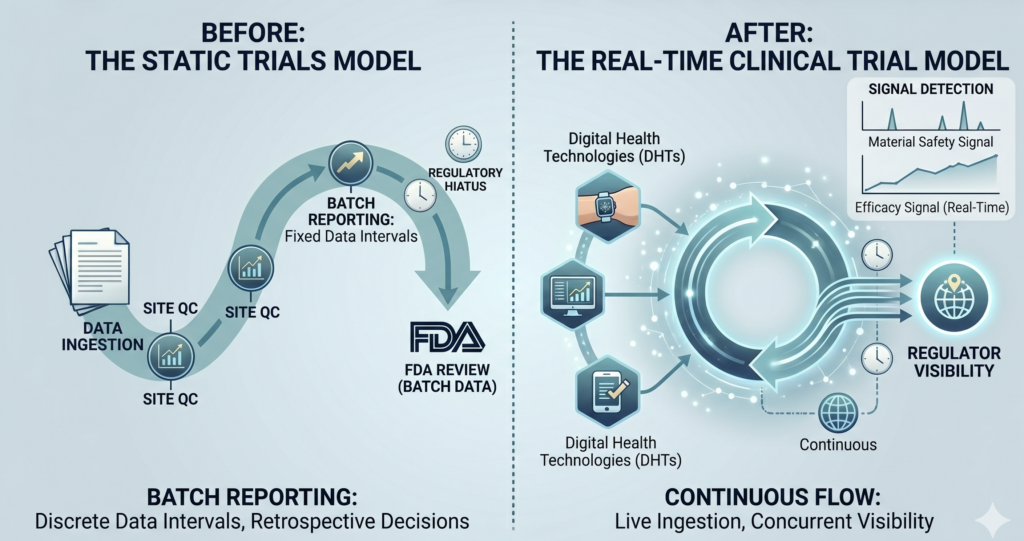
vs. Continuous Models
Fig 1: The Shift to Real-Time Clinical Trials: Static vs. Continuous Models
Call to Action
As we move toward the FDA’s finalized pilot selection in August 2026, we must ask ourselves: Are we simply moving faster, or are we building the infrastructure to move smarter?
I recommend that all site leads review their current data entry timelines. If your “time-to-entry” exceeds 24 hours, you are not yet ready for a Real-Time Clinical Trial.
Reference:

Leave a Reply